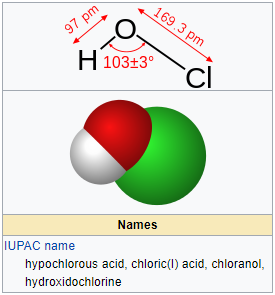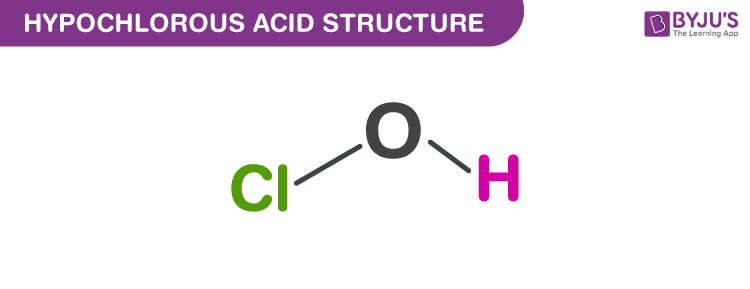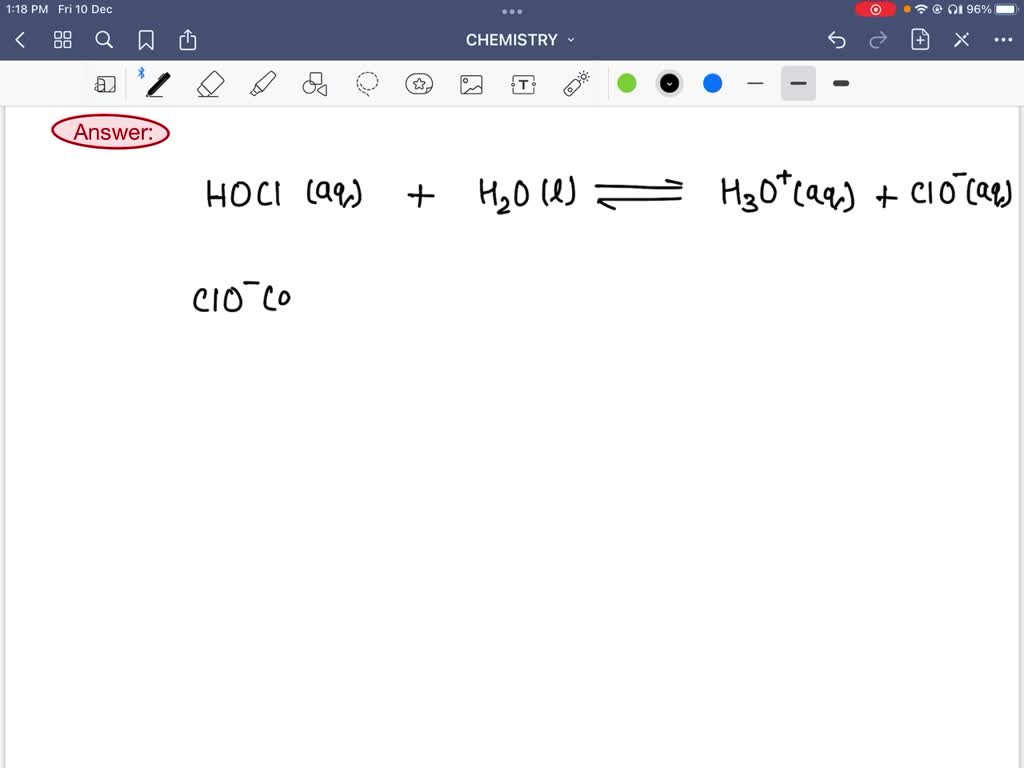
SOLVED: Hypochlorous acid ( HClO) is a weak acid. The conjugate base of this acid is the hypochlorite ion (ClO ). Wrtie a balanced equation showing the reaction of HClO with water.

Investigation of the Relationship Between H2O2 and HClO in Living Cells by a Bifunctional, Dual-ratiometric Responsive Fluorescent Probe | Analytical Chemistry
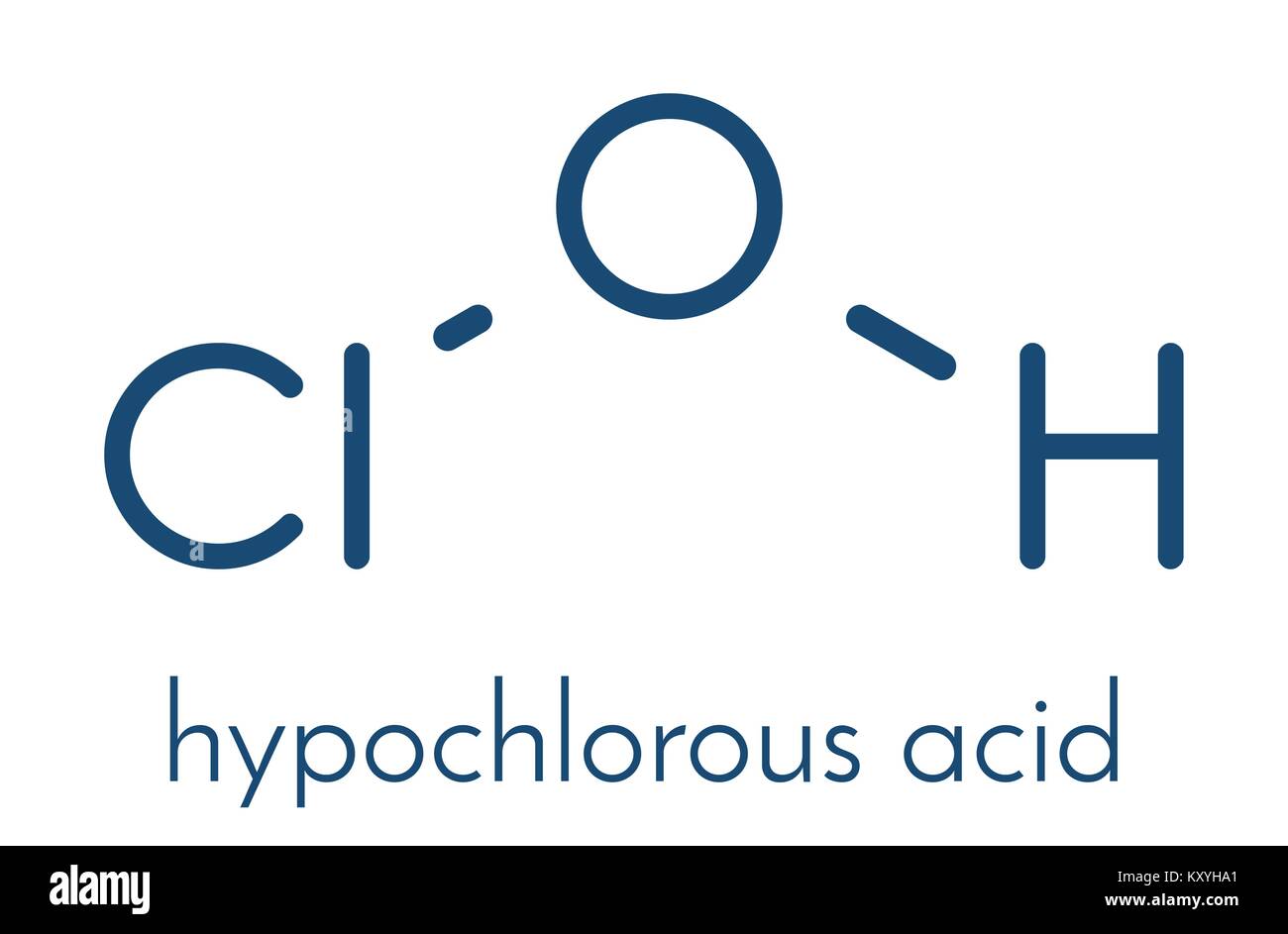
Hypochlorous acid (HClO) disinfectant molecule. Formed when chlorine is dissolved in water. Skeletal formula Stock Vector Image & Art - Alamy